Recombinant Human APP / Protease nexin-II Protein (Fc tag)
Amyloid precursor protein (APP) is a type I transmembrane protein expressed in many tissues and concentrated in the synapses of neurons, and is suggested as a regulator of synapse formation and neural plasticity. APP can be processed by two different proteolytic pathways. In one pathway, APP is cleaved by β- and γ-secretase to produce the amyloid-β-protein (Aβ, Abeta, beta-amyloid) which is the principal component of the amyloid plaques, the major pathological hallmark of Alzheimer’s disease (AD), while in the other pathway, α-secretase is involved in the cleavage of APP whose product exerts antiamyloidogenic effect and prevention of the Aβ peptide formation. The aberrant accumulation of aggregated beta-amyloid peptides (Abeta) as plaques is a hallmark of AD neuropathology and reduction of Abeta has become a leading direction of emerging experimental therapies for the disease. Besides this pathological function of Abeta, recently published data reveal that Abeta also has an essential physiological role in lipid homeostasis. Cholesterol increases Abeta production, and conversely A beta production causes a decrease in cholesterol synthesis. Abeta may be part of a mechanism controlling synaptic activity, acting as a positive regulator presynaptically and a negative regulator postsynaptically. The pathological accumulation of oligomeric Abeta assemblies depresses excitatory transmission at the synaptic level, but also triggers aberrant patterns of neuronal circuit activity and epileptiform discharges at the network level. Abeta-induced dysfunction of inhibitory interneurons likely increases synchrony among excitatory principal cells and contributes to the destabilization of neuronal networks. There is evidence that beta-amyloid can impair blood vessel function. Vascular beta-amyloid deposition, also known as cerebral amyloid angiopathy, is associated with vascular dysfunction in animal and human studies. Alzheimer disease is associated with morphological changes in capillary networks, and soluble beta-amyloid produces abnormal vascular responses to physiological and pharmacological stimuli.
Synonym(s) : AAA , APP , ABETA , ABPP , AD1 , APPI , CTF gamma , CVAP , PN2
Species(s) : Human
Protein Expression Host : Human Cells
Protein Construction : A DNA sequence encoding the human APP-751 isoform (NP_958816.1) (Met 1-Leu 669) was expressed with the C-terminal fused Fc region of human IgG1.
Predicted N-terminal amino acid : Leu 18
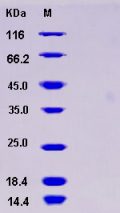
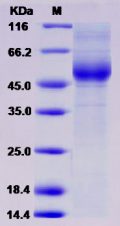
Molecular weight/mass : The recombinant human APP/Fc is a disulfide-linked homodimeric protein after removal of the signal peptide. The reduced monomer consists of 890 amino acids and predicts a molecular mass of 101 kDa. By SDS-PAGE under reducing conditions, the apparent molecular mass of rhAPP/Fc monomer is approximately 150-160 kDa due to the glycosylation.
Protein Construction : A DNA sequence encoding the human APP-751 isoform (NP_958816.1) (Met 1-Leu 669) was expressed with the C-terminal fused Fc region of human IgG1.
Formulation : Lyophilized from sterile PBS, pH 7.4
Purity : > 95 % as determined by SDS-PAGE
Endotoxin level : < 1.0 EU per μg of the protein as determined by the LAL method
Stability : Samples are stable for up to twelve months from date of receipt at -70°C
Shipping conditions : In general, recombinant proteins are provided as lyophilized powder which are shipped at ambient temperature.Bulk packages of recombinant proteins are provided as frozen liquid. They are shipped out with blue ice unless customers require otherwise.
Reconstitution : Please refer to the printed manual for detailed information.
Additional information
| Format | 10 ug, 20 ug |
|---|---|
| Species | Human |
Ask a question about Human APP / Protease nexin-II Protein (Fc tag) – PKSH031467
You must be logged in to post a review.



Reviews