Encor Mouse Monoclonal to Rhodopsin
Rhodopsin is the protein in the mammalian retina responsible for the light sensitivity of rod cells, which are responsible for vision in low light levels. Somewhat surprisingly, the rhodopsin protein turned out to be a typical member of the seven transmembrane G protein-coupled receptor (GPCR) superfamily. Whereas other GPCRs initiate signaling on binding a specific ligand, rhodopsin exists with a ligand already bound, specifically the vitamin A related substance retinal.
Retinal can exist in two isomeric forms, 11-cis and 11-trans retinal. In the dark, rhodopsin is associated with 11-cis retinal, but photons cause the 11-cis form to flip to the 11-trans form, and this causes an alteration in the structure of the rhodopsin making it catalytically active. Activated rhodopsin in turn activates the GTP binding protein G protein transducin by favoring the loss of GDP and the addition of GTP.
Transducin is a typical member of the family of heterotrimeric G proteins, and consists of an α and a βγ subunit. The α subunit is responsible for the GTP binding and the GTP bound form activates a phosphodiesterase (PDE) enzyme which hydrolyses cyclic GMP. This in turn increases the membrane potential of the rod cell and reduces the rate of synaptic signaling. So light stimulation actually results in a reduced rate of photoreceptor synaptic release. This information is transmitted through neurons of the retina to the visual centers of the brain (see review 1, 2).
Rhodopsin activity is shut off by phosphorylation under the influence of rhodopsin kinase, the activity of which results in binding of visual arrestin (a.k.a. arrestin-1 and S-antigen), which prevents rhodopsin from interacting with and activating more transducin molecules (3, 4). This basic signaling paradigm proved to be a prototype for understanding how other GPCRs function, as proteins similar to transducin, arrestin and rhodopsin kinase are found in these pathways.
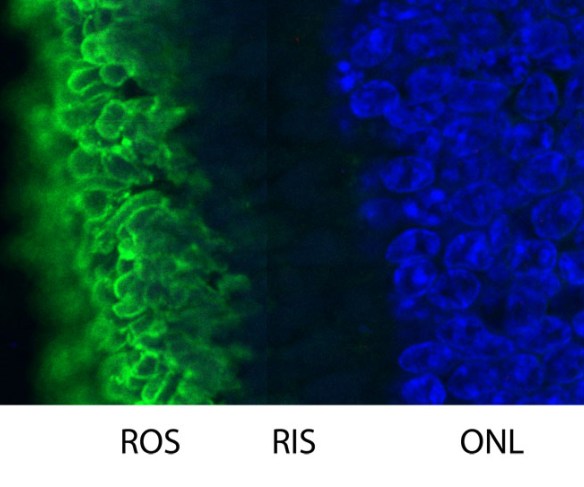
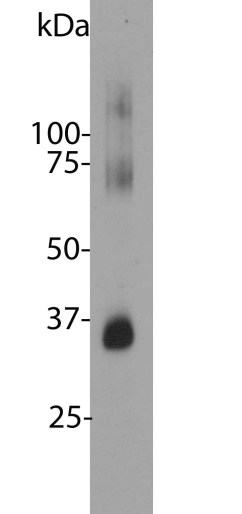
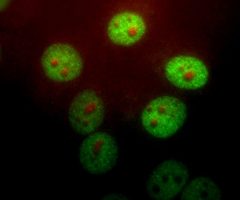
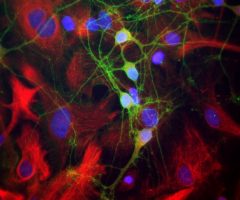
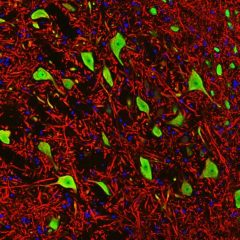
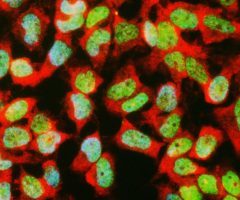
MCA-B630 was raised against bovine outer segment discs and shows convincing staining for rhodopsin both on western blots and on sections of retina. The protein is concentrated in rod outer segments, and the MCA-B630 epitope has been mapped to the amino-terminus and is therefore on the exterior side of the membrane. The antibody has been used successfully in fluorescence activated cell sorting (FACS). The HGNC name for this protein is RHO.
HGNC name(s) : RHO
Host : Mouse
Clonality : Monoclonal
ID : EnCor Biotechnology Rhodopsin B630
Reactivity : Human | Cow | Rat | Mouse | likely works in all mammals.
Isotype : IgG1
Conjugation : none
Immunogen : Bovine outer segment discs
Mass of detected protein : 35 kDa (higher MW aggregates detected on SDS-PAGE)
Uniprot ID : P61586
KGNC name : RHO
RRID # : AB_2572379
Purification : Affinity purified at 1 mg/mL
Storage : Shipped on ice. Store at 4°C. For long term storage, leave frozen at -20°C. Avoid freeze / thaw cycles.
Validated applications : WB | IF/ICC | IHC
Suggested Dilutions:
WB:1:5 000, ICC/IF and IHC: 1:1 000.
References :
1. Molday RS. Photoreceptor membrane proteins, phototransduction, and retinal degenerative disease. The Frienwald lecture. Invest Ophthalmol Vis Sci. 39:2491-513 (1998).
2. Yau,KW . Phototransduction Mechanism in Retinal Rods and Cones. The Frienwald lecture. Invest Ophthalmol Vis Sci. 35:9-32 (1994).
3. Wilden U, Hall SW, Kühn H. Phosphodiesterase activation by photoexcited rhodopsin is quenched when rhodopsin is phosphorylated and binds the intrinsic 48-kDa protein of rod outer segments. Proc Natl Acad Sci USA 83:1174-8 (1986).
4. Smith WC, Mc Dowell JH, Dugger DR, Miller R, Arendt A, Popp MP, Hargrave PA. Identification of regions of arrestin that bind to rhodopsin. Biochemistry Mar 38:2752-61 (1999).
Additional information
| Format | 50 ul, 100 ul, 500 ul |
|---|---|
| Supplier | |
| Host | Mouse |
| Clonality | Monoclonal |
| Reactivity | all mammals expected, Cow, Human, Mouse, Rat |
| Validated Applications | IHC, IF/ICC, WB |
| Conjugation | None |
| Isotype | IgG1 |

Reviews